|
Last Update:
Thursday November 22, 2018
|
| [Home] |
|
Volume 21 Issue 1 Pages 1 - 55 (July 2004) Citation: Evangelista, E. (2004) Change Of Partners In A Giant Otter Alpha Couple . IUCN Otter Spec. Group Bull. 21(1): 47 - 51 Change Of Partners In A Giant Otter Alpha Couple Emanuela Evangelista Associação Amazonia Ong, Conj. Residencial S.J. Taddeo Bl.7 Apt.304 Parque das Laranjeiras, Manaus - AM, Brasil. e-mail: eman_evan@hotmail.com
INTRODUCTION A population of giant otters (Pteronura brasiliensis) in the Xixuau Reserve in Roraima, Brazil, was studied from October 2000 to March 2003 in order to gather biological and ecological data. Five resident groups were followed during the study. According to the literature, a group of giant otters consists of a reproductive pair (alpha couple) and one or two litters (DUPLAIX, 1980; STAIB, 1995; CARTER and ROSAS 1997; ROSAS and de MATTOS, 2003). A change of partners occurred in one of the main study groups (M group) that had been observed for three consecutive dry seasons: (1) from October 2000 to March 2001, (2) from November 2001 to May 2002, (3) from October 2002 to March 2003. STUDY AREA The Xixuau Reserve (0º48.023'S, 61º33.476'W; altitude 30m a.s.l.) is located 500 km north-west of Manaus and consists mainly of primary tropical forest crossed by a black water river (River Jauperi) and many creeks. During the flood season, the water level usually rises up to 12m, flooding wide portions of the forest and sandy beaches along the watercourses. The area is virtually uninhabited, with a human population density of 0.04 persons per km2. METHODS Daily
surveys were conducted during the dry season by canoe to monitor
an area of approximately 60 km2. Riverbanks and lakes were patrolled
searching for indirect signs, such as campsites, tracks or dens, and
the positions recorded by GPS. The resident groups were followed at
a distance of 10-100 meters and filmed by a camcorder, the individuals
recognised by their throat markings. Observations from shelters were
achieved in the proximity of the dens at dawn and sunset, when the
behaviour of the individuals was recorded using the ad libitum sampling
where no specific constraints are put on what is recorded or when and
the observer write down anything that seems relevant or interesting
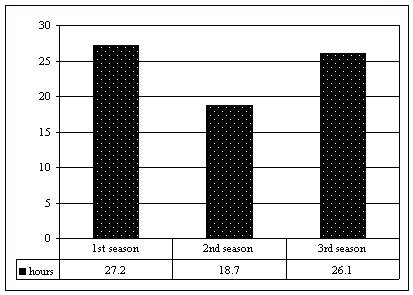
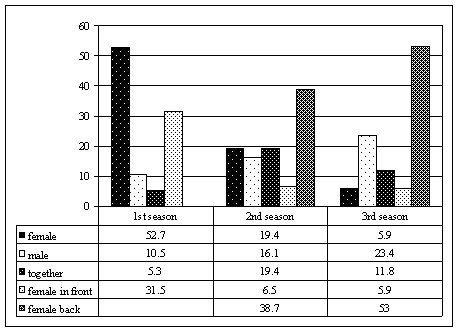
at the time. Between October 2000 and March 2003, the M group was sighted 99 times, with a total of over 71 hours of observation (Figure 1). When first seen, the group was composed of an alpha couple (named Eme and Moro) that gave birth to a cub (Dago) two months later. This composition (1:1:1) lasted up to March 2001. From January 2001, a solitary otter was sighted within the group's territory, once leaving a single untrampled scat on a latrine in use by the M group. The neck pattern of the newcomer was recorded and he was named Kappa. From the beginning of the second dry season, November 2001, the father Moro was no longer sighted and had been replaced by Kappa. The group remained stable in its new form and was observed in the same home range until May 2002. In October 2002, the new couple (Eme and Kappa) had a litter of two cubs (Juan and Zoe) and maintained this composition (1:1:3) until the end of the fieldwork. BEHAVIOURAL CHANGES OF THE ALPHA FEMALE During the first season of the study, when Eme had Moro as her partner, she used to play a dominant role, taking the front line when an intruder (i.e. the human observer) was sighted (Figure 2). Out of 37 sightings, the typical alarm behaviour (snorting, periscoping, charging) was recorded 19 times, 10 of which were performed by Eme alone (52,7%), 2 by Moro alone (10,5%), 1 by the two adults together (5,3%) and 6 by Eme in front and Moro behind (31,5%). During the second season, after the partner change, Eme tended to stay back, leaving the leadership role to Kappa. Out of 37 sightings, 31 alarms were recorded, 6 performed by Eme alone (19,4%), 5 by Kappa alone (16,1%), 6 by the two adults together (19,4%), 2 by Eme in front and Kappa behind (6,5%), and 12 by Kappa in front and Eme behind (38,7%). During the third season, after the new litter, Eme continued to show the same tendency. Out of 25 sightings, 17 alarms were recorded, 1 performed by Eme alone (5,9%), 4 by Kappa alone (23,4%), 2 by the two adults together (11,8%), 1 by Eme in front and Kappa behind (5,9%) and 9 by Kappa in front and Eme behind (53%).
DISCUSSION Changes in alpha couple composition of giant otter groups has never been recorded in the literature. In the present study, the alpha male disappeared and was replaced by another adult male. As the change of partner occurred in the rainy season, when field observation was not possible, it was not ascertained if the former male died or moved elsewhere or if the takeover was pacific or determined by fights. The killing of the immature infants or juveniles by conspecifics other than parents occurs in a variety of animal taxa, from invertebrates to vertebrates (EBENSPERGER, 1998). Among giant otters, however, only one case has been documented so far, where an adult male entered the den of a different group and cannibalised a cub (MOURAO and CARVALHO, 2001). Kappa adopted Moro's cub (Dago) and bred with the female only one year later. When Kappa was first seen within the group's territory Dago was about one month old, but when the complete transition occurred Dago's exact age was not known, though it is possible that the cub's age may have played a role in its survival. Clearly, more studies are needed to clarify whether infanticide or adoption is usual among giant otters. Defence seems to be cooperative in giant otter groups (CARTER and ROSAS, 1997). According to DUPLAIX (1980), the role of charging the intruders is usually undertaken by adult males, whereas STAIB (1995) reports two cases of alpha females guarding the group. The change reported in Eme's behaviour seems to be an adjustment to the different behaviour of the two partners, the second male (Kappa) being more aggressive than the first (Moro). The task of taking the front line during alarm behaviour might depend on several factors, not only sex but also age, experience, character of an individual, as well as group composition. Among many mammals, the frequency and intensity of maternal aggression towards intruders increases during late gestation and lactation (EBENSPERGER, 1998). During the present study, the female Eme did not demonstrate any increase in aggressiveness after the birth of the second litter. Acknowledgements - I am in debt to the Associação Amazonia (Manaus, Brazil), GEV (Modena, Italy) and Canon for logistic and financial support. Special thanks are due to the native guides Carlos Alberto Nascimento e Alexandre Soares for their important help during the fieldwork. I also thank Fernando Rosas, Giuseppe M. Carpaneto and Gália E. de Mattos who provided critical and insightful comments on the text and data analysis. REFERENCES Carter,
S.K., Rosas, F.C.W. 1997. Biology and conservation of the giant otter Pteronura
brasiliensis. Mammal Review 27, 1-26. Résumé :
Changement De Partenaires Au Sein D'un Couple Alpha De Loutres GeantesIl
existe peu de données sur le comportement social de la loutre
géante (Pteronura brasiliensis). Dans cette note, je
reporte le cas d'un changement de partenaires au sein d'un couple
alpha de loutres géantes, observé dans la réserve
de Xixuau à Roraima, au Brésil. Le mâle du couple
reproducteur disparut et fut remplacé par un autre mâle
adulte, aperçu au préalable sur le territoire du groupe.
Le jeune du couple alpha d'origine survécut à la transition
et fut adopté par le nouveau mâle. Le groupe ainsi reformé resta
stable et un an plus tard, le couple eut une portée de deux
jeunes. La fréquence des réactions d'alarme fut relevé pour
chaque individu et les résultats montrent que la femelle modifie
son comportement en fonction de celui de son partenaire. Resumen: Cambio En La Composicin
De Una Pareja Reproductiva De Nutria Gigante |
| [Copyright © 2006 - 2050 IUCN/SSC OSG] | [Home] | [Contact Us] |